In all types of solar cells the energy from light is used to separate charge. In dye-sensitized solar cells, an intensely colored molecule absorbs light and transfers an electron into a semiconducting material such as titanium dioxide. We are investigating the fundamental energy and charge transfer dynamics that govern light absorption and charge separation in zinc oxide-based dye-sensitized solar cells. The overarching goal of this research is to study the relationship between structure, energetics, and dynamics in a set of synthetically controlled donor-acceptor dyads. These studies will provide access to a deeper understanding of the light absorption and charge transfer steps that lie at the heart of DSSCs, thus enabling significant future advances in cell efficiencies.
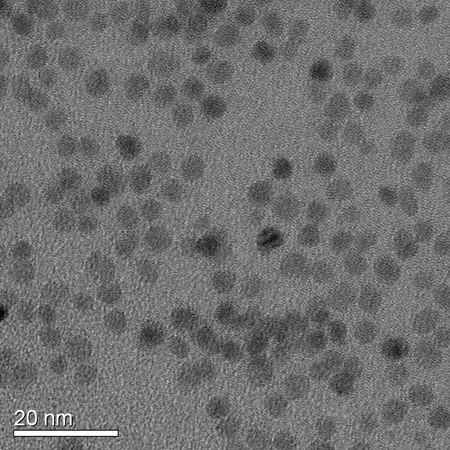
Zinc oxide has many properties that are similar to titanium dioxide, which is the most widely used metal oxide in dye cells. Some of zinc oxide's properties, such as it's electron mobility, are notably better, however, charge injection rates from the excited state of a dye into ZnO are slower. By synthetic modification of the dyes, we are able to manipulate their excited state potentials and the electronic coupling between the dye and the ZnO nanocrystal. By changing the diameter of the nanocrystals we can make use of quantum confinement to alter the energy and density of states of the ZnO conduction band. Coupled with detailed spectroelectrochemical studies of the dyes and detailed measurement of the charge transfer kinetics, we aim to understand the details of charge injection into ZnO.